From CLARITY Wiki
CLARITY is a technique developed in the Deisseroth lab at Stanford University.[1][2] The method is used to transform an intact tissue into an optically transparent and permeable hydrogel-hybridized form that can undergo immunostaining and high resolution 3-D imaging without damage to the sample. By clearing the tissue while preserving fine structural details, CLARITY provides a technique for obtaining high-resolution information from complex systems while maintaining the global perspective necessary to understand system function.
Contents |
Purpose
The difficulty of attaining detailed structural and molecular information from intact tissues has been a key challenge in studying biological systems. Both complete structural analysis (i.e. not reconstructed across tissue sections) and molecular phenotyping are desired to gain full insights into the relationships and functional mechanisms of biological systems. However, it has proven difficult to achieve both types of analysis in intact tissues. Methods that promote intact tissue imaging and structural analysis are incompatible with molecular phenotyping, while techniques that focus on molecular labelling require thin tissue sectioning which limits structural reconstruction. CLARITY addresses the need for studying intact systems with molecular-level resolution and a global scope by supporting both tissue imaging and molecular phenotyping of intact tissues.
CLARITY is designed to provide visualization of long-range cellular projections for three-dimensional tissue mapping of a variety of tissue types. Thus far, CLARITY has shown to be a viable method for intact tissue imaging of long-range projections, local circuit wiring, cellular relationships, subcellular structures, protein complexes, nucleic acids, and neurotransmitters. Intact tissues processed with CLARITY have also shown compatibility with in situ hybridization and antibody labelling techniques, even supporting multiple rounds of staining and de-staining.
Methodology
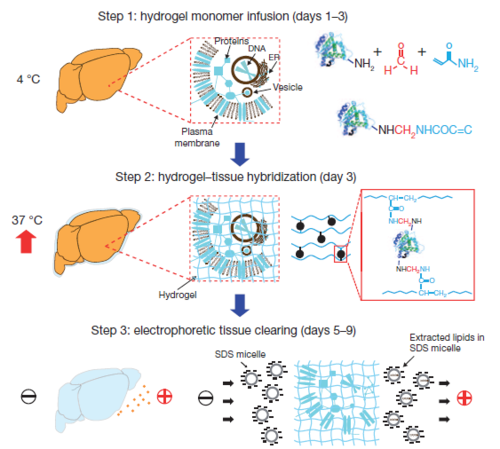
The major obstacle to studying intact biological systems is the presence of packed lipid bilayers within the tissue. By creating a diffusional barrier, the lipid membranes greatly limit the accessibility of molecular probes into the tissue. Additionally, the light-scattering that occurs at the lipid-aqueous interface causes intact tissues to be opaque and significantly restricts the imaging depth obtainable via light microscopy. However, the lipid membranes provide structural integrity and retain biomolecules in the tissue, so their removal would result in tissue damage and a profound loss of cellular and molecular information. CLARITY provides a method to remove the lipids while retaining other biomolecules within the intact tissue by first attaching the biomolecules to a hydrogel matrix to preserve their structural framework and then washing out the unattached lipids. The result is an optically transparent and macromolecule-permeable intact tissue-hydrogel hybrid with retained biological details. With the lipids removed, light and macromolecules can penetrate deep into the intact tissue, allowing three-dimensional imaging and molecular phenotyping without thin sectioning.
To create a hydrogel matrix within the intact tissue, the tissue is first infused at low temperature with a solution of small, hydrogel monomers including acrylamide and formaldehyde. Biomolecules within the tissue, such as proteins, nucleic acids, and small molecules, are covalently linked to the acrylamide monomers via reaction with formaldehyde. Using a thermally triggered initiator, the acrylamide is polymerized during high temperature incubation to form a hydrogel network crosslinked and held together by the attached biomolecules and a small chemical crosslinker. The resulting hydrogel-embedded tissue contains a physically supported structure of linked macromolecules throughout the physically-assembled packed lipid bilayers, which lack the functional groups necessary for chemical incorporation into the hydrogel mesh.
Once the hydrogel network is in place, the unattached lipid membranes can be removed without causing structural damage or loss to the intact embedded tissue. An ionic detergent, sodium dodecyl sulfide (SDS), that forms micelles in aqueous solution is used to collect the lipids and remove them from the tissue, leaving only the crosslinked biomolecules inside a swollen hydrogel matrix. An electric field can be applied to actively transport the SDS micelles through the tissue and expedite the lipid extraction process. Following lipid removal, the optically transparent and porous hydrogel-tissue hybrid can undergo immunostaining and/or whole-tissue three-dimensional imaging for high resolution analysis of the preserved fine structural details and molecular phenotypes in a global context.
Advantages and Disadvantages
Advantages
- No damage or thin sectioning required to visualize whole intact tissue samples
- Allows marking and visualization of long-range projections and subcellular structures
- Allows multiple rounds of molecular phenotyping
- Applicable to multiple tissue types and sizes
Disadvantages
- Multiple-step process that takes place over several days/weeks
- Immunostaining is time-consuming for thicker tissue samples
- High start-up and consumable material costs
Tissue Samples
Although originally developed for brain tissue, CLARITY can be performed on a variety of tissue types. Challenges may arise from tissues that are more fibrous (limited porosity). Tissues that contain light-scattering elements other than lipids, such as pigments, may require additional clearing mechanisms to remove those elements.
Tissues that have been successfully cleared using CLARITY include:
- Mouse brain
- Human brain slices
- Mouse spinal cord[3]
References
- ↑ K Chung, J Wallace, S-Y Kim, S Kalyanasundaram, AS Andalman, TJ Davidson, JJ Mirzabekov, KA Zalocusky, J Mattis, AK Denisin, S Pak, H Bernstein, C Ramakrishnan, L Grosenick, V Gradinaru, and K Deisseroth. Structural and molecular interrogation of intact biological systems. Nature (2013) 497: 332-337.
- ↑ K Chung and K Deisseroth. CLARITY for mapping the nervous system. Nature Methods (2013) 10(6): 508-513.
- ↑ M-D Zhang, G Tortoriello, B Hsueh, R Tomer, L Ye, N Mitsios, L Borgius, G Grant, O Kiehn, M Watanabe, M Uhlen, J Mulder, K Deisseroth, T Harkany, and TGM Hokfelt. Neuronal calcium-binding proteins 1/2 localize to dorsal root ganglia and excitatory spinal neurons and are regulated by nerve injury. PNAS (2014) 111(12): 1149-1158.